Ion bonding hydrogen bonding dipole dipole
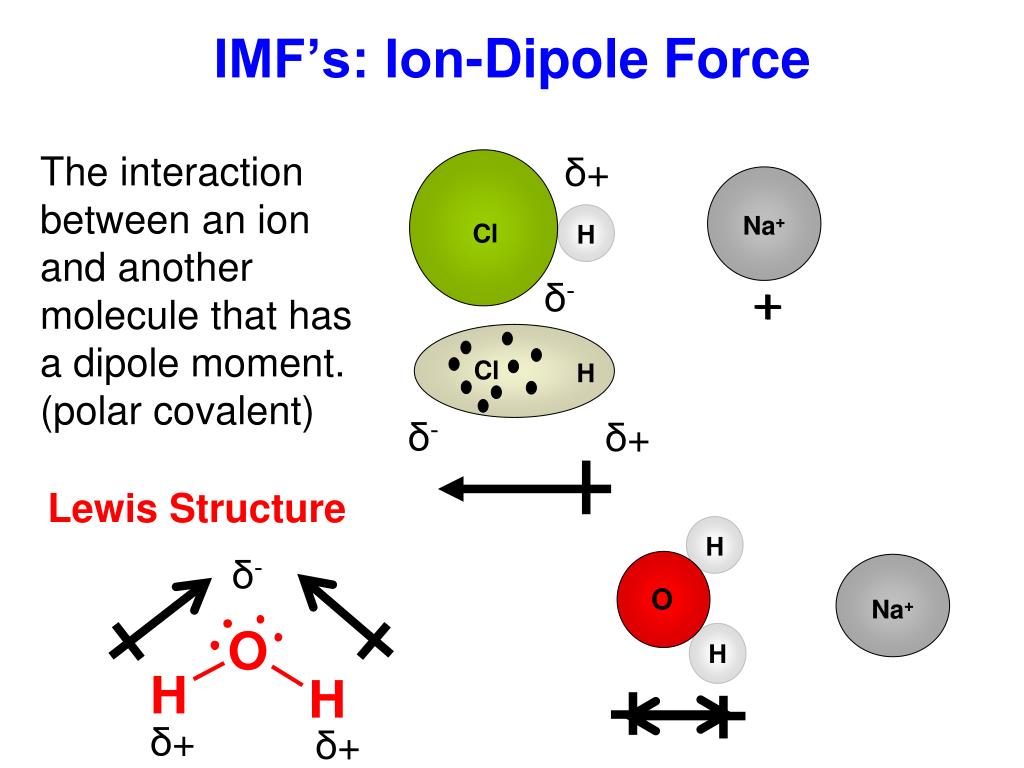
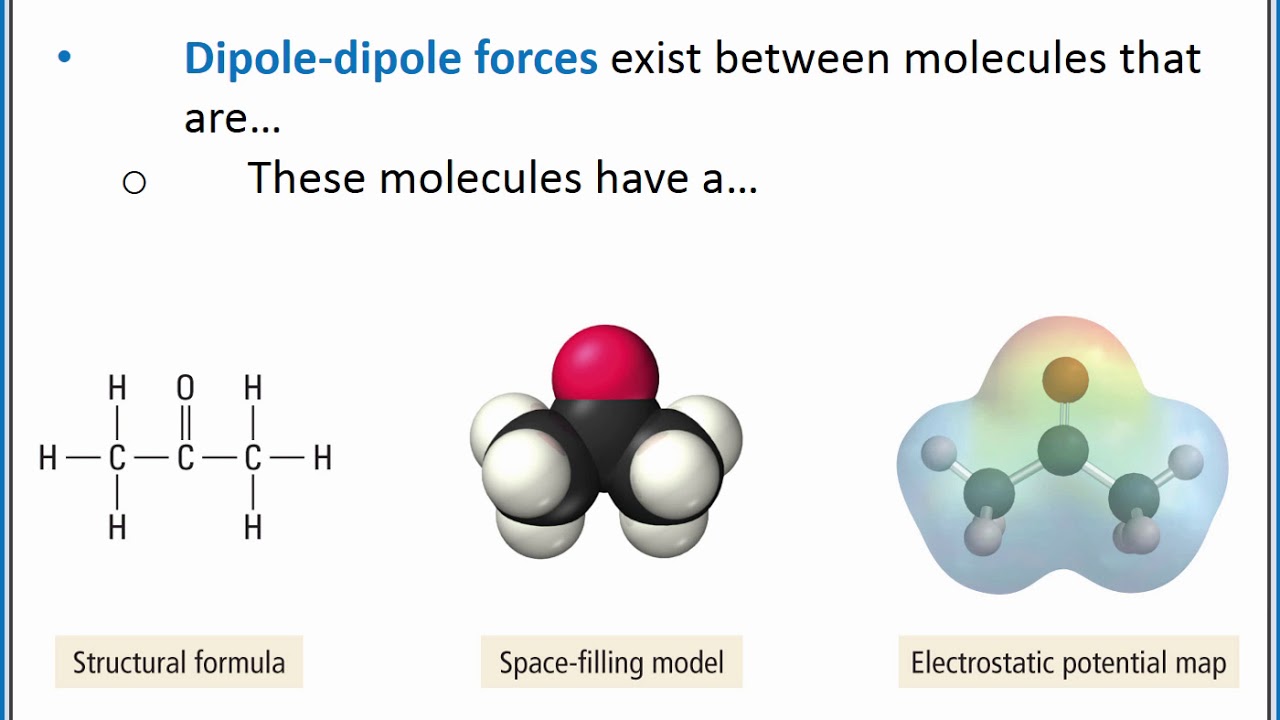
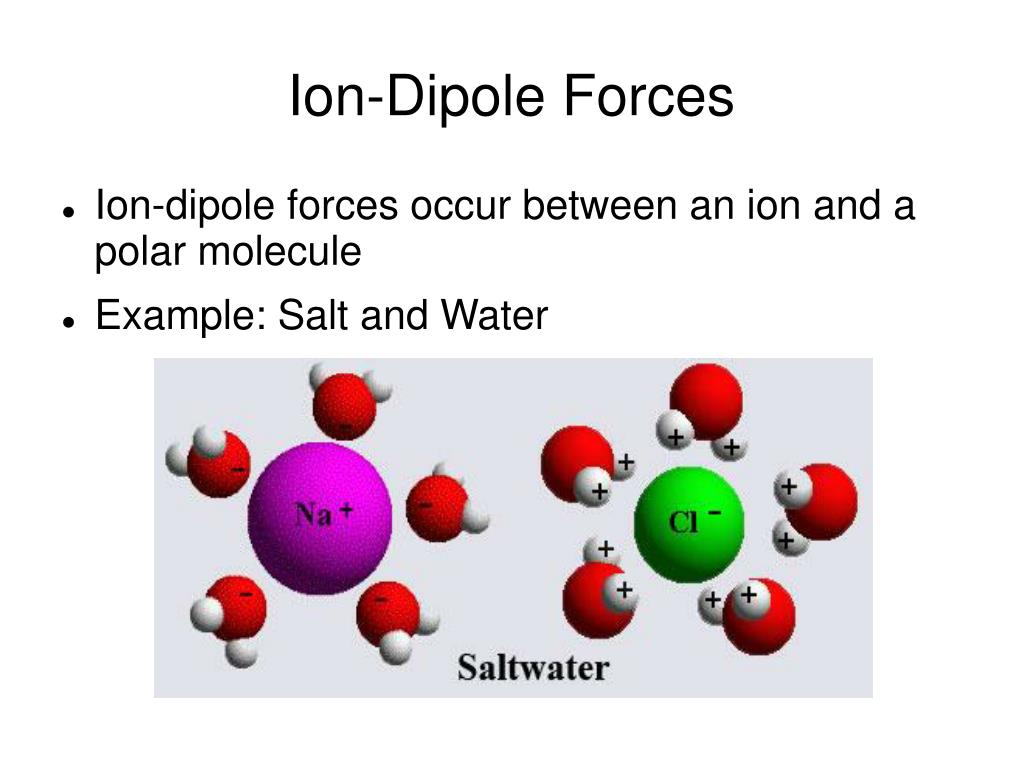
The attraction forces that arise between the slightly positive hydrogen atom of one molecule and the slightly negative chlorine atom of another molecule are known as dipole-dipole interaction. HCl has the dipole-dipole interaction and London dispersion forces present in between its molecules.ĭue to the electronegativity difference between hydrogen (2.2) and chlorine (3.16), a slight positive charge develops on the hydrogen atom while the chlorine atom acquires a slight negative charge. What types of intermolecular forces are present in HCl? In this article, you will learn everything you need to know about the intermolecular forces in HCl.
Hey Readers!!! Welcome to another fresh article on techiescientist.

It is used in the production of a number of inorganic compounds, in the pickling of steel, in pH control and neutralization reactions, etc. It is also found as a component of gastric acid in the stomach of humans as well as some other animals. In the industries, hydrochloric acid is prepared by reacting hydrogen chloride with water. it contains one atom of hydrogen and one atom of chlorine. It is denoted by the chemical formula HCl i.e. Hydrochloric acid is a colorless, pungent-smelling liquid.
